Popular on EntSun
- From Speech Therapy to 300+ Episodes: Brother Marcus Turns His Voice Into a Movement Launching a 24/7 Inspiration Radio Network on Day 100 of the Year - 472
- PlanetAI Nature Space (PNS), certificadora Europea, lanza su plataforma EUDR-PNS Ready basada en IA, satélites y trazabilidad blockchain - 148
- Raquel Riley Thomas' AOAGWLLC and Hazel Simpson's Sis to Sis Productions Ink Major Co-Production Deal, Launching Strategic Creative Alliance - 133
- Pacific Emblem Company Launches "Happy 250th Birthday America" Collection and Proud Supporter of the Gary Sinise Foundation - 132
- Century City Alumnae Chapter Of Delta Sigma Theta Sorority, Inc. Presents The 2026 Entertainment Career Summit At Emerson College Los Angeles - 131
- Charging Into the $30 Billion Heart Failure Market with Late-Stage Momentum, Breakthrough Data, & Strong Financial Backing: Cardiol Therapeutics $CRDL - 130
- Acuvance Appoints Sandeep Sabharwal to Board of Directors, Strengthening Leadership to Support Continued Platform Growth - 128
- Bold Beauty Project Celebrates Anniversary with Collaborative Exhibition at FIU's Miami Beach Urban Studios - 126
- Matter Studio Gallery Presented "Evolving Matter," a Pop-Up Exhibition Examining Material Transformation and Contemporary Form - 124
- Architect of Neurodiversity Will Lead the First U.S. Team of Autistic Children to the "Genius Cup" in Hiroshima, Japan, in 2027 - 115
Similar on EntSun
- Why Athletic Recovery Begins in the Nervous System
- 62% of Gen X have no estate planning documents — Trust & Will research identifies "the Sandwich Gap"
- $10 Million Annual Revenue Merger, Profitable Partner in AI Powered Specialty Automotive Sales Projected to Scale Above $200M: Stock Symbol: NWPG
- Virginia Moving Company Nearly Doubles Customer Calls in Two Weeks After Switching to CARL — the Bold New Alternative to WordPress
- RAS AP Consulting Launches Vendor Master File & Payment Controls Assessment for NACHA Phase 2 Compliance
- Strategic Talent Associates Launches THE ALIGNED RESET™
- Calvetta Phair Founder & CEO Earns AOPA Foundation Flight Training Scholarship, Inspiring a New Generation of STEM Dreamers in Underserved Communities
- Karen D. Gentry Announces New Book Focused on Relationships and Personal Growth
- New plusOne Research Finds the Orgasm Gap Is a 30-Point Chasm — and Confirms It Isn't Biology
- CCHR Report Links 145 Violent Incidents to Psychiatric Drug Exposure, Urges National Oversight and Action
FDA Meeting Indicates a pivotal development that could redefine the treatment landscape for suicidal depression via NRx Pharmaceuticals: $NRXP
EntSun News/11084680
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP) $NRXP: FDA Alignment, 70,000-Patient Real-World Data, and a Debt-Free Balance Sheet Position NRXP for Transformational 2026 Catalysts
MIAMI - EntSun -- In a pivotal development that could redefine the treatment landscape for suicidal depression, NRx Pharmaceuticals (N A S D A Q: NRXP) has emerged from a high-level, in-person Type C meeting with the U.S. Food and Drug Administration with what management believes is a clear path toward New Drug Application (NDA) submission for NRX-100 (preservative-free ketamine)—supported by both prior clinical trials and an unprecedented real-world dataset exceeding 65,000 patients.
For investors, the implications are substantial: regulatory clarity, expanding indications, strategic partnerships, a strengthened balance sheet, and a global ketamine market estimated at $750 million annually—with no FDA-approved drug currently indicated specifically for suicidal ideation.
FDA Type C Meeting Signals Regulatory Momentum
NRXP's meeting was attended by leadership from the FDA Division of Psychiatry Products and the Center for Drug Evaluation and Research (CDER)—a strong signal of institutional engagement at the highest levels.
Key takeaways:
This broader indication meaningfully expands the addressable patient population and commercial potential.
NRX-100 has already received Fast Track Designation for the treatment of acute suicidality in depression and bipolar depression.
70,000 Patients: Real-World Data at Scale
NRXP licensed data from more than 70,000 U.S. patients treated with IV ketamine or nasal S-ketamine for depression and suicidal ideation.
Preliminary analysis of a 20,000-patient subset demonstrated:
Notably, there is currently no FDA-approved medication specifically indicated for suicidal ideation, with Electroconvulsive Therapy (ECT) remaining the primary intervention.
More on EntSun News
If FDA alignment continues, NRX-100 could become the first drug positioned specifically for this indication under Accelerated Approval.
KETAFREE™: First Preservative-Free Ketamine
NRXP has applied for the proprietary name KETAFREE™, designed to be the first preservative-free ketamine formulation submitted for approval in this setting.
Given existing clinical familiarity with ketamine, the transition from generic compounded products to an FDA-reviewed, preservative-free branded formulation presents a compelling commercialization thesis.
Dual Strategy: Drug Development + Clinic Network Expansion
In parallel, NRXP announced a joint offering with neurocare Group AG to create a nationwide network of integrated neuroplastic care clinics targeting:
The model integrates:
Recent publications demonstrate:
NRXP plans to integrate neurocare clinics with HOPE Therapeutics and leverage an installed base of 400+ Apollo® TMS machines nationwide, creating a scalable service revenue channel alongside drug commercialization.
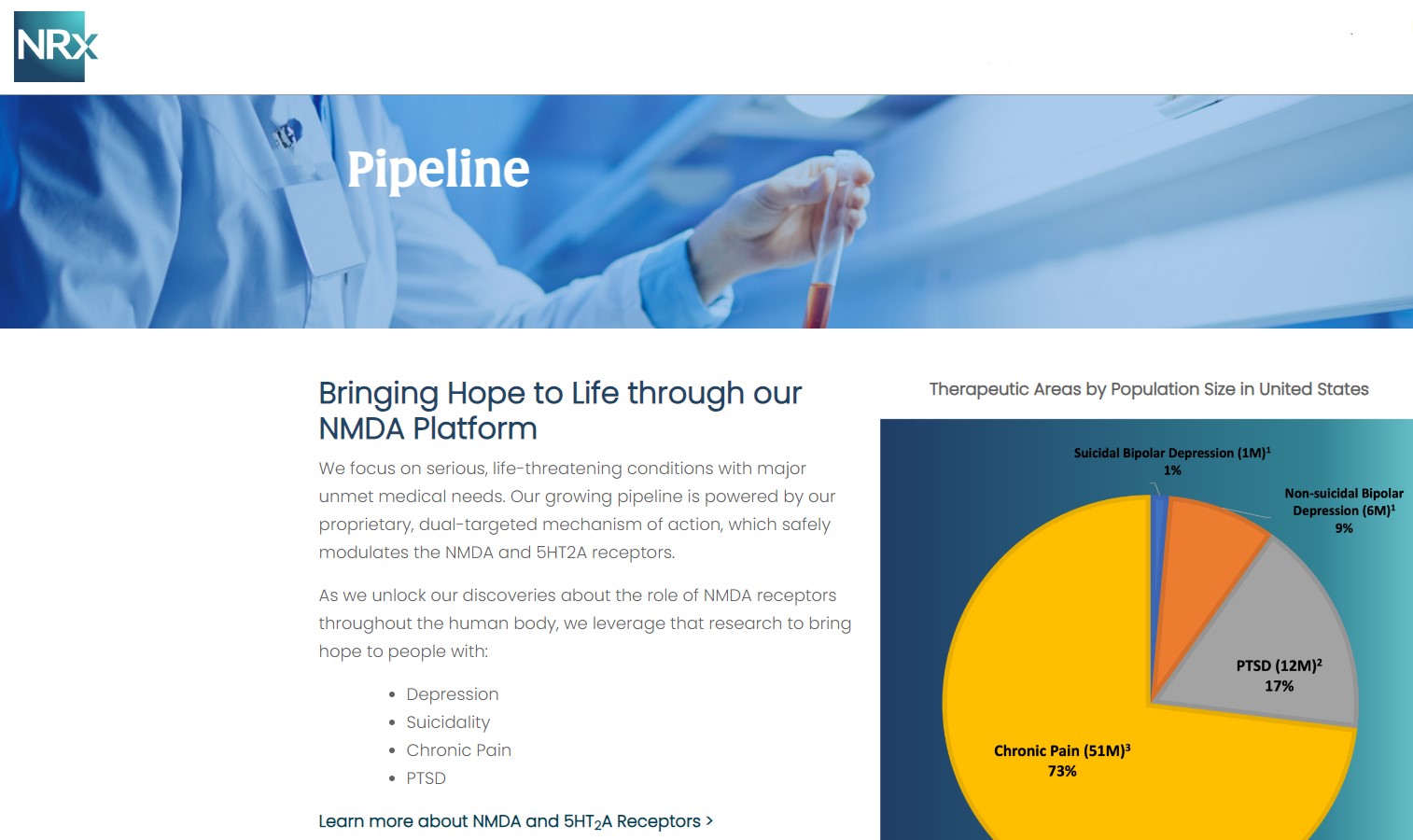
NRX-101: Breakthrough Therapy with Expanded Market Potential
Beyond NRX-100, NRXP is advancing NRX-101, an FDA-designated Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain.
Key attributes:
With projections suggesting 1 million Americans annually may receive TMS by 2030, NRX-101 could capture a significant share of a rapidly expanding neurostimulation market.
NRXP has partnered with Alvogen Pharmaceuticals for development and marketing of NRX-101 in suicidal bipolar depression.
Debt-Free Balance Sheet: Strategic Reset for Growth
More on EntSun News
In December, NRXP eliminated its remaining $5.4 million in balance sheet debt via equity conversion—with no warrants or toxic provisions.
This clean capital structure positions the company for:
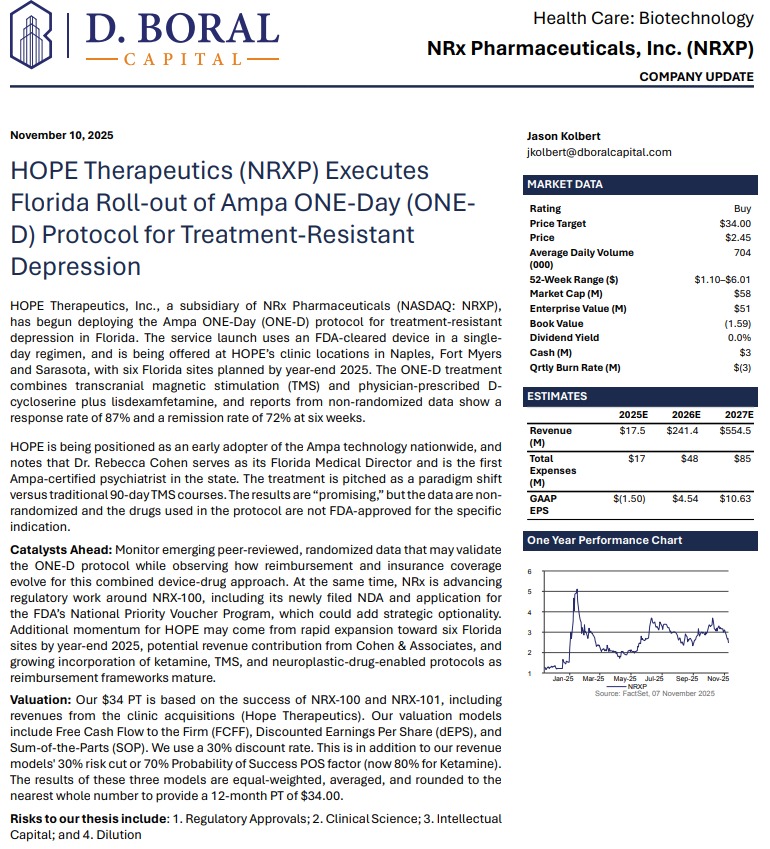
Analyst Coverage: $34 Price Target
Investment firm D. Boral has issued a Buy rating with a $34 price target, citing regulatory progress, expanded indications, and commercial optionality.
For a company operating at the intersection of FDA reform, real-world evidence utilization, and neuroplastic treatment innovation, this represents a high-conviction thesis on both regulatory and market catalysts.
The Macro Opportunity
According to CDC data, more than 13 million Americans seriously consider suicide each year.
Yet there is no FDA-approved drug indicated specifically for suicidal ideation.
NRXP is positioning itself to:
This is not a single-asset story—it is a platform strategy targeting one of the largest and most urgent unmet needs in psychiatry.
Investment Thesis Snapshot
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Media Contact
Company Name: NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Contact Person: Matthew Duffy, Chief Business Officer
Company Website: https://www.nrxpharma.com/
Email: mduffy@nrxpharma.com
Phone: (484) 254-6134
Home Country: United States
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
For investors, the implications are substantial: regulatory clarity, expanding indications, strategic partnerships, a strengthened balance sheet, and a global ketamine market estimated at $750 million annually—with no FDA-approved drug currently indicated specifically for suicidal ideation.
FDA Type C Meeting Signals Regulatory Momentum
NRXP's meeting was attended by leadership from the FDA Division of Psychiatry Products and the Center for Drug Evaluation and Research (CDER)—a strong signal of institutional engagement at the highest levels.
Key takeaways:
- Path to NDA based on existing adequate and well-controlled trial data
- Submission supported by 65,000+ patient Real-World Evidence (RWE) dataset
- No additional nonclinical studies required
- No bridging studies needed for preservative-free formulation
- Opportunity to seek broader indication: Treatment-Resistant Depression (TRD) with suicidality—not just acute suicidality
This broader indication meaningfully expands the addressable patient population and commercial potential.
NRX-100 has already received Fast Track Designation for the treatment of acute suicidality in depression and bipolar depression.
70,000 Patients: Real-World Data at Scale
NRXP licensed data from more than 70,000 U.S. patients treated with IV ketamine or nasal S-ketamine for depression and suicidal ideation.
Preliminary analysis of a 20,000-patient subset demonstrated:
- Rapid resolution of depression
- Rapid reduction in suicidal ideation
- Clinical outcomes consistent with prior randomized trials
- Favorable comparison to currently approved therapies
Notably, there is currently no FDA-approved medication specifically indicated for suicidal ideation, with Electroconvulsive Therapy (ECT) remaining the primary intervention.
More on EntSun News
- Nayarit's Jungle Coast Redefines Luxury Travel on Mexico's Pacific Now More Accessible Than Ever
- RUNWAY® Magazine Launches Official Merchandise Collection Amid Devil Wears Prada 2 release
- Debut Novel HAMMER by Joe Betar Delivers a Relentless, High-Stakes Thriller
- $10 Million Annual Revenue Merger, Profitable Partner in AI Powered Specialty Automotive Sales Projected to Scale Above $200M: Stock Symbol: NWPG
- Virginia Moving Company Nearly Doubles Customer Calls in Two Weeks After Switching to CARL — the Bold New Alternative to WordPress
If FDA alignment continues, NRX-100 could become the first drug positioned specifically for this indication under Accelerated Approval.
KETAFREE™: First Preservative-Free Ketamine
NRXP has applied for the proprietary name KETAFREE™, designed to be the first preservative-free ketamine formulation submitted for approval in this setting.
Given existing clinical familiarity with ketamine, the transition from generic compounded products to an FDA-reviewed, preservative-free branded formulation presents a compelling commercialization thesis.
Dual Strategy: Drug Development + Clinic Network Expansion
In parallel, NRXP announced a joint offering with neurocare Group AG to create a nationwide network of integrated neuroplastic care clinics targeting:
- Depression
- PTSD
- Bipolar Depression
- Autism
- Traumatic Brain Injury
The model integrates:
- Transcranial Magnetic Stimulation (TMS)
- Ketamine and neuroplastic drugs
- Hyperbaric oxygen therapy
- Supportive psychotherapy
Recent publications demonstrate:
- 87% clinical response
- 72% remission
in treatment-resistant depression when combining TMS with neuroplastic therapies.
NRXP plans to integrate neurocare clinics with HOPE Therapeutics and leverage an installed base of 400+ Apollo® TMS machines nationwide, creating a scalable service revenue channel alongside drug commercialization.
NRX-101: Breakthrough Therapy with Expanded Market Potential
Beyond NRX-100, NRXP is advancing NRX-101, an FDA-designated Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain.
Key attributes:
- Composition-of-matter patent protection globally
- Combines D-cycloserine + low-dose lurasidone
- Designed to mitigate hallucination risk seen with DCS alone
- Newly amended IND to include augmentation of TMS
With projections suggesting 1 million Americans annually may receive TMS by 2030, NRX-101 could capture a significant share of a rapidly expanding neurostimulation market.
NRXP has partnered with Alvogen Pharmaceuticals for development and marketing of NRX-101 in suicidal bipolar depression.
Debt-Free Balance Sheet: Strategic Reset for Growth
More on EntSun News
- Really Cool Music Releases Its First-Ever Latin Single "Esa Hondureña"
- Weirding Way Media Launches "What, No F**kin' Ziti?", A New Podcast Devoted To HBO's The Sopranos
- Memories Unleashed at North Coast Rep Variety Nights
- When Myth Meets Biology: A New Take on Vampire Lore in Namtar: The Night Plague
- Cat Rodriguez Launches Second Season Golf, Redefining Vintage and Secondhand Golf Apparel for Women
In December, NRXP eliminated its remaining $5.4 million in balance sheet debt via equity conversion—with no warrants or toxic provisions.
This clean capital structure positions the company for:
- NDA filing preparation
- Clinic expansion
- Commercial scale-up
- Potential strategic partnerships
Analyst Coverage: $34 Price Target
Investment firm D. Boral has issued a Buy rating with a $34 price target, citing regulatory progress, expanded indications, and commercial optionality.
For a company operating at the intersection of FDA reform, real-world evidence utilization, and neuroplastic treatment innovation, this represents a high-conviction thesis on both regulatory and market catalysts.
The Macro Opportunity
According to CDC data, more than 13 million Americans seriously consider suicide each year.
Yet there is no FDA-approved drug indicated specifically for suicidal ideation.
NRXP is positioning itself to:
- Be first-to-label in suicidal depression
- Expand into broader treatment-resistant depression
- Integrate drug + device neuroplastic therapies
- Monetize both pharmaceutical and clinic service channels
This is not a single-asset story—it is a platform strategy targeting one of the largest and most urgent unmet needs in psychiatry.
Investment Thesis Snapshot
- ✅ Fast Track designation for NRX-100
- ✅ 70,000-patient real-world dataset
- ✅ Clear FDA path toward NDA
- ✅ Broader proposed indication
- ✅ Breakthrough Therapy designation (NRX-101)
- ✅ Expanding TMS augmentation indication
- ✅ Strategic partnership with neurocare
- ✅ Debt-free balance sheet
- ✅ Global ketamine market opportunity
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Media Contact
Company Name: NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Contact Person: Matthew Duffy, Chief Business Officer
Company Website: https://www.nrxpharma.com/
Email: mduffy@nrxpharma.com
Phone: (484) 254-6134
Home Country: United States
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Source: CorporateAds
Filed Under: Business
0 Comments
Latest on EntSun News
- BoroBased Hosts "Back to the Boros"
- Club Moda USA Unveils 2026 Cruise & Resort Wear Collection Ahead of Memorial Day Weekend
- Food Journal Magazine Unveils Its Latest 'Best of Los Angeles' Culinary Discoveries
- Boston Industrial Solutions Launches Natron® 717S Series: A New Flexible UV LED Ink for Ricoh GH2220 Printheads
- 5 Things NYC Courier Services Won't Tell You About How Same-Day Delivery Actually Works
- Tropidelic Links Up With International Reggae Star Collie Buddz and Eli Mac for Feel-Good Breakthrough Single "Follow Your Nature"
- Save 15 Percent on Florida Keys Accommodations with KeysCaribbean's 'Advance Purchase Rate Discount'
- Zma/odflix Studio Announces Epic Fantasy Thriller Wizard Hunter 1: The Hunt For Evangelion Crowley
- Atelier 411 Studios and Columbus Fashion Council Present Red Carpet Experience at Gateway Film Center for The Devil Wears Prada 2
- Hazel E Celebrates Birthday with Luxury "Goddess" Yacht Experience in Marina del Rey
- Lecture/Performance on Jewish Magicians in Juneau on Monday, May 4, 2026
- Joseph Neibich sits down with Bold Jounrey (aka Joseph Nybyk)
- AI Suite 360 Launches Done-For-You AI Implementation to Rescue SMBs from the "Frankenstein Tax"
- Live Comedy Comedy Battle Mountain Thursday May 7
- CX Network Releases Report on the Best AI Support Tools for SaaS Companies 2026
- Outlier Pest Season Hits Willamette Valley as Mild Winter Drives Early Surge in Ant and Rodent Activity
- 9th Annual Baton Rouge Soul Food Festival Announces Talent Lineup, Contests and Sponsors
- Lokal Media House Wins Web Excellence Award for Black Plumbing Redesign
- 8XBET International Sports Technology Enhances Digital Fan Experience in West Africa
- Lick Expands Flavored Massage Oil Collection with 10 New Indulgent Cream-Inspired Scents