Popular on EntSun
- Century City Alumnae Chapter Of Delta Sigma Theta Sorority, Inc. Presents The 2026 Entertainment Career Summit At Emerson College Los Angeles - 167
- RAS AP Consulting Advances to RFP Stage in Heidelberg Materials' SAP Vendor & Customer Master Data Modernization Initiative - 144
- Advanced TeleSensors Appoints AgeTech Innovator Tiffany Wey, MBA as Vice President of Sales & Marketing - 130
- Slipaway Food Truck Park & Marina to host Kentucky Derby watch party and Cinco de Mayo celebration - 121
- Most Americans Choose Their Water Brand Because of Its Natural Source — Yet Fewer Than 3 in 10 Understand What Spring Water Actually Is - 116
- A Hidden Magical World Awaits in Ashley Gayheart's Upcoming Young Adult Fantasy, Rosewood Academy: The Awakening - 115
- Lecture/Performance on Jewish Magicians in Juneau on Monday, May 4, 2026 - 114
- CCHR Report Links 145 Violent Incidents to Psychiatric Drug Exposure, Urges National Oversight and Action - 106
- Calvetta Phair Founder & CEO Earns AOPA Foundation Flight Training Scholarship, Inspiring a New Generation of STEM Dreamers in Underserved Communities - 104
- New research identifies The Discovery Gap: Seven in 10 Americans say travel is no longer just about getting away - 102
Similar on EntSun
- Collectibles EvoRelic Celebrates Stellar 4.8-Star Customer Rating
- Pediatrician Launches "Confessions of a Detective Doctor" Children's Book Series
- T. Jones Group's Cameron Jones Serves as Judge for the 2026 CHBA National Awards for Housing Excellence
- $29.8 Million Record Setting Q1 with Boosted Annual Guidance to $160 Million for Expanding Pre-Owned Boat Dealer: Off The Hook YS, Inc. N Y S E: OTH
- All About Technology Celebrates 25 Years of Bridging Detroit's Digital Divide
- iatroX surpasses 500,000 clinical queries and expands specialist exam coverage
- SRK Collective Media Group Launches with a Modern Approach to Media, Authority Building, and Cultural Visibility
- MSBG Corporation Acquires GridWatch US Telemetry Automation System
- KT Medical Staffing Expands Concierge Nursing and Private Duty Nursing Services in Orange County
- T. Jones Group Named Finalist Across Multiple Categories at the 2026 Georgie Awards
NRx Pharmaceuticals Launches Breakthrough One-Day Treatment Clinic in Florida as FDA Pathway and Clinical Data Strengthen Growth Outlook; $NRXP
EntSun News/11086376
87% Clinical Response From Non-Invasive Neuroplastic Therapy Positions $NRXP at the Forefront of Next-Generation Mental Health Treatment. NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
MIAMI - EntSun -- NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP) is accelerating its push to transform the treatment landscape for severe mental health disorders with the launch of a new interventional psychiatry clinic in Palm Beach, Florida offering a one-day treatment protocol for depression and PTSD.
The company's innovative approach combines ketamine-based therapies, transcranial magnetic stimulation (TMS), neuroplastic medications, hyperbaric oxygen therapy, and physician-led psychotherapy—a multi-modal treatment platform designed to deliver rapid clinical improvement for patients suffering from treatment-resistant conditions.
Published clinical research has demonstrated an 87% clinical response rate in patients receiving short-term neuroplastic therapy protocols for treatment-resistant depression, highlighting the transformative potential of this approach.
With millions of patients struggling with severe depression and PTSD, NRx Pharmaceuticals is positioning itself as a next-generation leader in neuropsychiatric medicine and interventional mental health care.
New Palm Beach Clinic Expands HOPE Therapeutics Network
On March 9, the company announced the opening of its Palm Beach, Florida clinic, now accepting patients for advanced treatment of depression and post-traumatic stress disorder.
The facility becomes the latest location within the company's HOPE Therapeutics clinic network, which is focused on delivering cutting-edge neuroplastic treatments nationwide.
NRXP's treatment model integrates:
Pilot programs incorporating these therapies have demonstrated up to a 90% return-to-function rate in patients suffering from depression and PTSD.
Additionally, scientific studies have shown TMS may improve memory and cognitive function in patients with Alzheimer's disease and traumatic brain injury, expanding potential future treatment applications.
The Palm Beach location also benefits from the company's nationwide partnership with neurocare Group AG, bringing advanced non-invasive psychiatric technologies to a growing network of treatment centers.
Breakthrough Drug Development Pipeline Targets Massive Unmet Need
NRx Pharmaceuticals is simultaneously advancing a powerful clinical pipeline focused on serious central nervous system disorders.
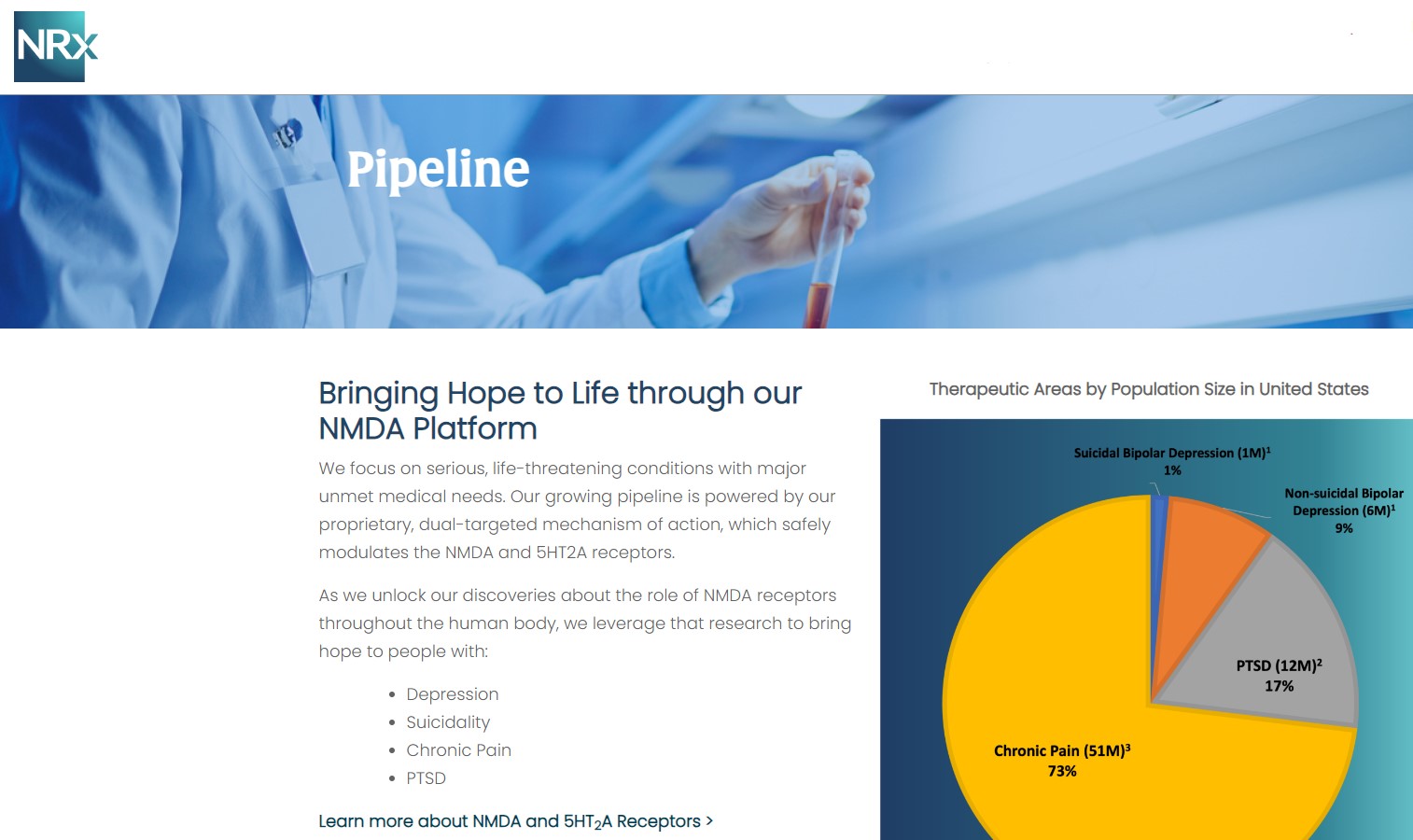
The company's lead program, NRX-101, has been granted FDA Breakthrough Therapy designation for the treatment of suicidal treatment-resistant bipolar depression and chronic pain.
This therapy is designed to address the needs of more than 13 million Americans who seriously consider suicide each year, according to data from the Centers for Disease Control and Prevention.
More on EntSun News
NRX-101 is being developed in collaboration with Alvogen Pharmaceuticals and may also offer a non-opioid treatment pathway for chronic pain, representing an additional large market opportunity.
FDA Meeting Confirms Path Toward New Drug Approval for NRX-100 Ketamine Therapy
In February, NRXP announced the successful completion of an in-person Type C meeting with the U.S. Food and Drug Administration at the agency's headquarters.
The meeting included leadership from:
Based on guidance received, the company believes it now has a clear pathway to filing a New Drug Application (NDA) for NRX-100, its preservative-free intravenous ketamine formulation.
The application will be supported by:
Importantly, the FDA advised the company that no additional nonclinical data or bridging studies would be required to review the preservative-free formulation.
NRXP plans to seek a broader treatment indication, targeting patients with treatment-resistant depression associated with suicidality rather than only the most acute suicidal subset.
Real-World Evidence From 70,000 Patients Strengthens Regulatory Case
NRXP has licensed Real-World Evidence data from more than 70,000 patients in the United States treated with intravenous ketamine or nasal S-ketamine for depression and suicidal ideation.
This dataset is expected to play a pivotal role in the company's regulatory submission for Accelerated Approval of NRX-100 under FDA Fast Track designation.
Preliminary analysis of a 20,000-patient subset demonstrated rapid resolution of depression symptoms and suicidal ideation following ketamine treatment.
These results were found to be consistent with prior randomized clinical trials and compare favorably with currently approved therapies.
Notably, no medications are currently approved specifically to treat suicidal ideation, leaving electroconvulsive therapy as the only FDA-approved intervention for acute cases.
If approved, NRX-100 could represent a major breakthrough in psychiatric medicine.
Partnership With neurocare Group Expands Nationwide Treatment Network
NRXP recently announced a strategic collaboration with neurocare Group AG to develop a nationwide network of clinics delivering integrated neuroplastic therapies.
The combined treatment platform includes:
Recent scientific publications show this integrated approach has achieved 87% response rates and 72% remission rates in patients with treatment-resistant depression.
More on EntSun News
The program will leverage more than 400 installed Apollo® TMS systems nationwide, accelerating the rollout of advanced mental health treatments.
Future research initiatives between the two companies will also explore applications in:
World-Class Clinical Leadership Strengthens Innovation Strategy
NRXP recently appointed Professor Joshua C. Brown, MD, PhD, as its Chief Medical Innovation Officer.
Dr. Brown is widely recognized as a pioneer in Transcranial Magnetic Stimulation research and currently serves as:
He has extensive research experience in neuroplasticity and psychiatric recovery, as well as work advancing TMS treatment strategies for military personnel, veterans, and first responders.
The company's clinical leadership team includes physicians trained at leading institutions such as:
Debt-Free Balance Sheet Sets Stage for Accelerated Growth
In December, NRXP eliminated all remaining balance sheet debt through a strategic equity conversion totaling $5.4 million, leaving the company with a clean capital structure.
The debt-free balance sheet positions the company to pursue:
Heading into 2026, the company believes it is positioned for significant operational and clinical growth.
Analyst Coverage Sees Significant Upside Potential
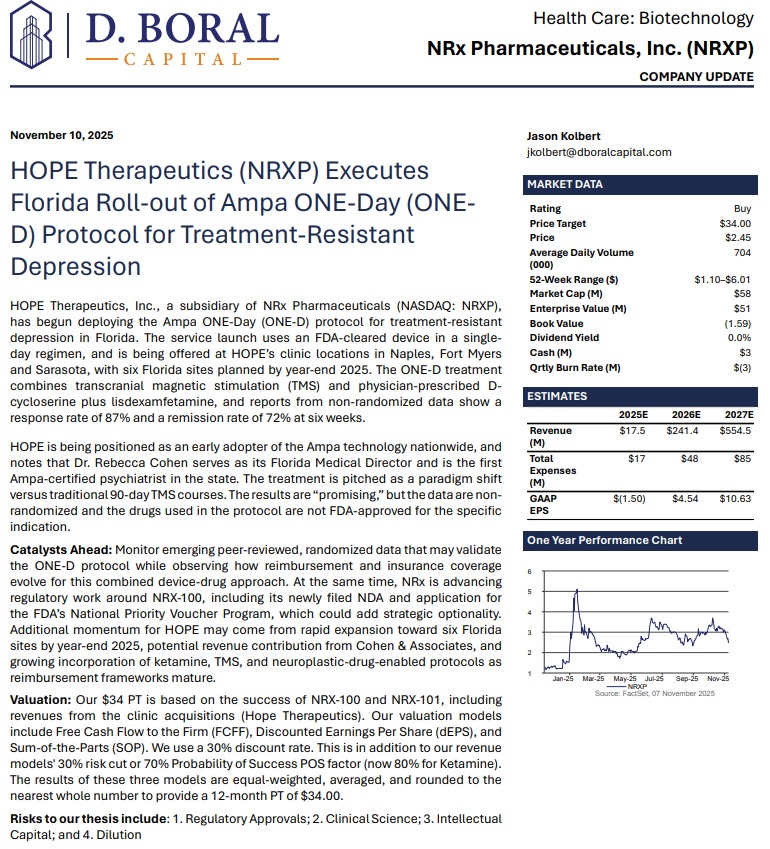
Independent analyst firm D. Boral Capital has issued a Buy rating on NRXP with a $34 price target, reflecting the potential value of the company's clinical pipeline, treatment network expansion, and regulatory progress.
The full analyst report can be accessed at:
https://www.nrxpharma.com/wp-content/uploads/2025/11/HOPE-Therapeutics-NRXP-Executes-Florida-Roll-out-of-Ampa-O.pdf
Positioned at the Intersection of Breakthrough Psychiatry and a $750 Million Ketamine Market
NRx Pharmaceuticals is developing KETAFREE™, a proprietary preservative-free ketamine formulation intended to improve safety and clinical usability.
The current global generic ketamine market is estimated at approximately $750 million annually, presenting a meaningful commercial opportunity for innovative formulations and delivery models.
By integrating drug development, clinical treatment networks, and neuroplastic therapy technologies, NRXP is building a comprehensive platform aimed at transforming the treatment of depression, PTSD, and other severe neurological conditions.
About NRx Pharmaceuticals
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP) is a clinical-stage biopharmaceutical company developing therapies based on its NMDA platform for the treatment of central nervous system disorders including suicidal bipolar depression, chronic pain, and PTSD.
Company Contact
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Matthew Duffy – Chief Business Officer
Phone: (484) 254-6134
Email: mduffy@nrxpharma.com
Company Website:
https://www.nrxpharma.com/
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
The company's innovative approach combines ketamine-based therapies, transcranial magnetic stimulation (TMS), neuroplastic medications, hyperbaric oxygen therapy, and physician-led psychotherapy—a multi-modal treatment platform designed to deliver rapid clinical improvement for patients suffering from treatment-resistant conditions.
Published clinical research has demonstrated an 87% clinical response rate in patients receiving short-term neuroplastic therapy protocols for treatment-resistant depression, highlighting the transformative potential of this approach.
With millions of patients struggling with severe depression and PTSD, NRx Pharmaceuticals is positioning itself as a next-generation leader in neuropsychiatric medicine and interventional mental health care.
New Palm Beach Clinic Expands HOPE Therapeutics Network
On March 9, the company announced the opening of its Palm Beach, Florida clinic, now accepting patients for advanced treatment of depression and post-traumatic stress disorder.
The facility becomes the latest location within the company's HOPE Therapeutics clinic network, which is focused on delivering cutting-edge neuroplastic treatments nationwide.
NRXP's treatment model integrates:
- Ketamine and other neuroplastic medications
- Transcranial Magnetic Stimulation (TMS)
- Hyperbaric oxygen therapy
- Physician-guided psychotherapy
Pilot programs incorporating these therapies have demonstrated up to a 90% return-to-function rate in patients suffering from depression and PTSD.
Additionally, scientific studies have shown TMS may improve memory and cognitive function in patients with Alzheimer's disease and traumatic brain injury, expanding potential future treatment applications.
The Palm Beach location also benefits from the company's nationwide partnership with neurocare Group AG, bringing advanced non-invasive psychiatric technologies to a growing network of treatment centers.
Breakthrough Drug Development Pipeline Targets Massive Unmet Need
NRx Pharmaceuticals is simultaneously advancing a powerful clinical pipeline focused on serious central nervous system disorders.
The company's lead program, NRX-101, has been granted FDA Breakthrough Therapy designation for the treatment of suicidal treatment-resistant bipolar depression and chronic pain.
This therapy is designed to address the needs of more than 13 million Americans who seriously consider suicide each year, according to data from the Centers for Disease Control and Prevention.
More on EntSun News
- Lux Boutique Launches Stylish "For the Pets" Collection for Fashion-Forward Pet Lovers
- Pediatrician Launches "Confessions of a Detective Doctor" Children's Book Series
- Integrated Maintenance Platforms Are Transforming Aircraft Operations
- T. Jones Group's Cameron Jones Serves as Judge for the 2026 CHBA National Awards for Housing Excellence
- 40 Year Anniversary California Strawberry Festival Poster Revealed; Artist to Autograph Posters
NRX-101 is being developed in collaboration with Alvogen Pharmaceuticals and may also offer a non-opioid treatment pathway for chronic pain, representing an additional large market opportunity.
FDA Meeting Confirms Path Toward New Drug Approval for NRX-100 Ketamine Therapy
In February, NRXP announced the successful completion of an in-person Type C meeting with the U.S. Food and Drug Administration at the agency's headquarters.
The meeting included leadership from:
- The FDA Division of Psychiatry Products
- The FDA Office of Neuroscience
- The FDA Center for Drug Evaluation and Research (CDER)
Based on guidance received, the company believes it now has a clear pathway to filing a New Drug Application (NDA) for NRX-100, its preservative-free intravenous ketamine formulation.
The application will be supported by:
- Data from well-controlled clinical trials conducted with the U.S. National Institutes of Health
- Large Real-World Evidence datasets
- Confirmatory clinical analyses
Importantly, the FDA advised the company that no additional nonclinical data or bridging studies would be required to review the preservative-free formulation.
NRXP plans to seek a broader treatment indication, targeting patients with treatment-resistant depression associated with suicidality rather than only the most acute suicidal subset.
Real-World Evidence From 70,000 Patients Strengthens Regulatory Case
NRXP has licensed Real-World Evidence data from more than 70,000 patients in the United States treated with intravenous ketamine or nasal S-ketamine for depression and suicidal ideation.
This dataset is expected to play a pivotal role in the company's regulatory submission for Accelerated Approval of NRX-100 under FDA Fast Track designation.
Preliminary analysis of a 20,000-patient subset demonstrated rapid resolution of depression symptoms and suicidal ideation following ketamine treatment.
These results were found to be consistent with prior randomized clinical trials and compare favorably with currently approved therapies.
Notably, no medications are currently approved specifically to treat suicidal ideation, leaving electroconvulsive therapy as the only FDA-approved intervention for acute cases.
If approved, NRX-100 could represent a major breakthrough in psychiatric medicine.
Partnership With neurocare Group Expands Nationwide Treatment Network
NRXP recently announced a strategic collaboration with neurocare Group AG to develop a nationwide network of clinics delivering integrated neuroplastic therapies.
The combined treatment platform includes:
- Transcranial Magnetic Stimulation
- Ketamine and other neuroplastic drugs
- Hyperbaric oxygen therapy
- Structured psychotherapy programs
Recent scientific publications show this integrated approach has achieved 87% response rates and 72% remission rates in patients with treatment-resistant depression.
More on EntSun News
- Kinetics Academy of Dance Brings "Into the Jungle" to The Westport
- Captiva Cruises launches Memorial Day weekend sunset cruise to North Captiva Island
- The AI Direction Deficit: TripleTen Study Finds Staff Get Told to Use AI — But Not Trained to Use It
- The Greatest Film Director You Never Saw Comes to Blu-Ray from BayView Entertainment
- MetroLagoons announces summer camps at Epperson Lagoon
The program will leverage more than 400 installed Apollo® TMS systems nationwide, accelerating the rollout of advanced mental health treatments.
Future research initiatives between the two companies will also explore applications in:
- Bipolar depression
- Autism spectrum disorders
- Traumatic brain injury
World-Class Clinical Leadership Strengthens Innovation Strategy
NRXP recently appointed Professor Joshua C. Brown, MD, PhD, as its Chief Medical Innovation Officer.
Dr. Brown is widely recognized as a pioneer in Transcranial Magnetic Stimulation research and currently serves as:
- President of the Clinical TMS Society
- Editor-in-Chief of the Transcranial Magnetic Stimulation Journal
He has extensive research experience in neuroplasticity and psychiatric recovery, as well as work advancing TMS treatment strategies for military personnel, veterans, and first responders.
The company's clinical leadership team includes physicians trained at leading institutions such as:
- Harvard University
- Johns Hopkins University
- Georgetown University
Debt-Free Balance Sheet Sets Stage for Accelerated Growth
In December, NRXP eliminated all remaining balance sheet debt through a strategic equity conversion totaling $5.4 million, leaving the company with a clean capital structure.
The debt-free balance sheet positions the company to pursue:
- Clinic network expansion
- Drug development milestones
- Regulatory submissions
- Strategic partnerships
Heading into 2026, the company believes it is positioned for significant operational and clinical growth.
Analyst Coverage Sees Significant Upside Potential
Independent analyst firm D. Boral Capital has issued a Buy rating on NRXP with a $34 price target, reflecting the potential value of the company's clinical pipeline, treatment network expansion, and regulatory progress.
The full analyst report can be accessed at:
https://www.nrxpharma.com/wp-content/uploads/2025/11/HOPE-Therapeutics-NRXP-Executes-Florida-Roll-out-of-Ampa-O.pdf
Positioned at the Intersection of Breakthrough Psychiatry and a $750 Million Ketamine Market
NRx Pharmaceuticals is developing KETAFREE™, a proprietary preservative-free ketamine formulation intended to improve safety and clinical usability.
The current global generic ketamine market is estimated at approximately $750 million annually, presenting a meaningful commercial opportunity for innovative formulations and delivery models.
By integrating drug development, clinical treatment networks, and neuroplastic therapy technologies, NRXP is building a comprehensive platform aimed at transforming the treatment of depression, PTSD, and other severe neurological conditions.
About NRx Pharmaceuticals
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP) is a clinical-stage biopharmaceutical company developing therapies based on its NMDA platform for the treatment of central nervous system disorders including suicidal bipolar depression, chronic pain, and PTSD.
Company Contact
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Matthew Duffy – Chief Business Officer
Phone: (484) 254-6134
Email: mduffy@nrxpharma.com
Company Website:
https://www.nrxpharma.com/
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Source: CorporateAds
Filed Under: Business
0 Comments
Latest on EntSun News
- MetroLagoons announces Memorial Day weekend festivities
- EDC Weekend Comedy Special Featuring Don Barnhart & Friends — Use Promo Code FRIEND for 50% Off
- N Y S E: OTH Off The Hook YS Is Building a Vertically Integrated Marine Empire — And Investors Are Starting to Notice
- Concierge Title Agency Merges with Independence Title, Inc. to Deliver an Expanded Concierge Closing Experience Across South Florida
- Grow My Security Company Launches Next-Generation Website and Expands Strategic Marketing Solutions for the Security Industry
- $4.8M in Contracted AI Revenue with Projections of $30M Over 6-12 Months for Diversified AI Software and Platform-Based Services Provider XMax Inc
- Michelangelo's Great Secret Hiding in Plain Sight
- Virginia Marchese's Paradox: A Nation Still Deciding Who Belongs Examines Race, Migration, Law, and America's Unfinished Struggle for Equality
- From Blank Page to Published Book
- Larry R. Wasion's Jump Gate III RoadMaker Blends Cutting-Edge Sci-Fi with High-Stakes Space Exploration and Complex Technologies
- American Mensa and Davidson Institute Join Forces To Strengthen Support for Profoundly Gifted Youth
- 16th Annual Art Of Brooklyn Film Fest Returns June 1-10 with 55 New Indies
- 360 Sound And Vision Releases The Ubiquitous Compact Disc and America's Most Deadly UFO Encounters
- SpeedyIndex Rolls Out Automated API for Mass URL Verification, Solving the Backlink Blind Spot for SEO Agencies
- DJ Serving Grand Rapids, Detroit, and Ann Arbor Areas Walks Clients Through the Process
- KLEKT Announces Appointment of Jay Kimpton to Board of Directors
- Michigan Attorney General Closed FGM Licensing Investigations Months Before Federal Case Ended, Records Show
- Mensa Foundation Event Reframes Brain Health for Every Age
- Robert Woeger Announces New Christian Movie Review Of "I Am Living Proof" Documentary Movie
- DLT Resolution, Inc. (Stock Symbol: DLTI) Expands Into the $224 Billion Life Settlements Market While Accelerating Telecom Growth Across Canada